Presented at ESCMID Global 2026
Susanne Paukner1, Stephen Hawser2, David R. Cameron1
1. Debiopharm International SA, Switzerland
2. IHMA Europe Sàrl, Monthey, Switzerland
BACKGROUND
Afabicin is a novel first-in-class, narrow-spectrum antibacterial selectively targeting Staphylococcus spp. with the potential for sparing the gut microbiota. Afabicin is a prodrug that is converted to its active form afabicin desphosphono which inhibits the FabI enzyme in bacteria, crucially disrupting their fatty acid synthesis.
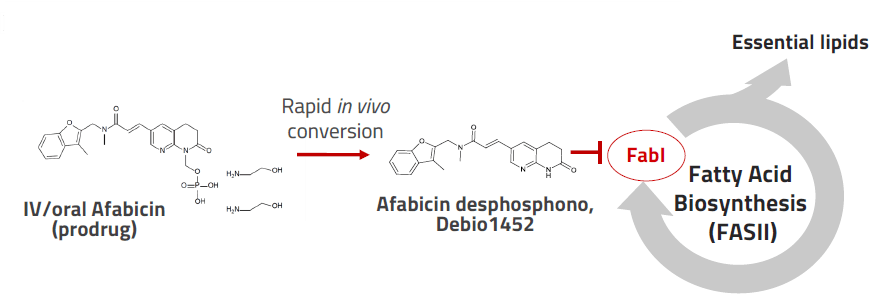
Promising safety and efficacy results have been obtained in Phase 2 trials in acute bacterial skin and skin structure infections (ABSSSI) and bone and joint infections (BJI), following intravenous and oral administrations.
This surveillance study investigated the antibacterial activity of afabicin desphosphono and comparators against recent clinical Staphylococcus spp. isolates collected globally during 5 years of surveillance.
